Cell Line Profile MDCK
(ECACC catalogue number 85011435)
Cell line history
The parental MDCK cell line was derived in 1958 from the kidney of a normal adult Cocker Spaniel by SH Madin and NB Darby Jnr. The cell line was established alongside cell lines from other mammalian species.
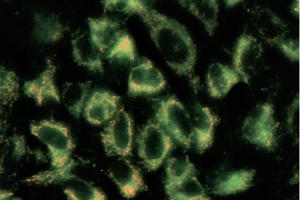
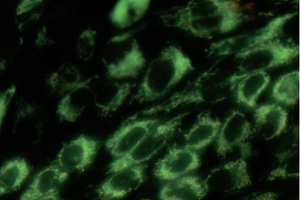
ECACC 85011435 MDCK cells stained with the mitochondrial dye (JC-1).
The image on the left shows red mitochondrial fluorescence and the image on the right shows loss of red fluorescence and increased cytoplasmic diffusion of green monomeric fluorescence due to the loss of mitochondrial membrane potential following exposure of the cells to carbonyl cyanide m-chlorophenyl hydrazone (CCCP) a chemical inhibitor of oxidative phosphorylation. Loss of mitochondrial membrane potential is a hallmark of apoptosis. (Molecular Devices ImageXpress® Nano Automated Imaging System (20X magnification) with CellReporterXpress Image Acquisition and Analysis Software - courtesy Simon Lydford, Molecular Devices).
Key characteristics
The MDCK cell lines are widely used as epithelial models as they have clear apico-basolateral polarity and form well defined tight junctions. They have the advantage of rapid cell growth and are well suited to use in advanced (e.g. confocal) microscopic techniques. Subsequently many studies into epithelial cell trafficking, cell polarity and tight junctions have relied heavily on the use of MCDK cells and its sub-clones1. If allowed to reach confluence on conventional tissue culture plastics MDCK monolayers exhibit “doming”; where the cells become polarised and actively transport solutes through the epithelial monolayer to the plastic below.
A commonly used method for evaluating the competence of in vitro epithelial barriers grown on Transwell® systems is to measure the trans epithelial electrical resistance (TEER) across the cell monolayer as first demonstrated by Cereijido et al in 19782. JM Arthur in 2000 showed that the MDCK parental cell line is heterogeneous and made up of populations of cells with different resistive and transport properties. This heterogeneity helps explain different characteristics of the cell line’s numerous subclones3.
MDCK I (ECACC 00062106) has been shown to have high (TEER) values indicating the cell line expresses “tight” junctions, however MDCK II (ECACC 0006210) cells were obtained from higher passage cultures and display lower TEER values, indicating that the junctions are more “leaky”. These differences stem from differences in the expression of junctional complexes in the cells4 .
Applications
MDCK cells were initially used in virology, they are permissive and support the growth of influenza virus5 making them suitable for virology research, diagnostics, and vaccine production6 The MDCK SIAT1 sub-clone (ECACC 05071502) expresses high levels of sialic acid making it especially useful in infection studies7.
More recently MDCK cells have been used extensively to help model, image and explore the properties of mammalian epithelia in vitro1.
Culture tips
MDCK cells express tight junctions at confluence it is therefore essential to keep growing stocks sub-confluent or they become very difficult to trypsinise. It is very important to give MDCK cell monolayers two or more washes with calcium and magnesium free PBS before trypsinisation, keeping the PBS on the cells for several minutes before aspiration. The cells may take longer than expected to detach.
|
Related cell lines |
ECACC catalogue number |
Description |
|---|---|---|
|
DoCI1 (S+L-) |
Canine kidney, Moloney Sarcoma Virus infected |
|
|
MDCK |
Canine Cocker Spaniel kidney |
|
|
MDCK-Protein Free |
Canine Cocker Spaniel kidney |
|
|
MDCK-SIAT1 |
Canine Cocker Spaniel Kidney Sialic Acid Over Expression |
|
|
MDCK I |
Canine Cocker Spaniel kidney |
|
|
MDCK II |
Canine Cocker Spaniel Kidney |
Key references
-
Dukes, J., Whitley, P. & Chalmers, A. The MDCK variety pack: choosing the right strain. BMC Cell Biol. 12, 43 (2011).
-
Cereijido, M., Robbins, E. S., Dolan, W. J., Rotunno, C. A. & Sabatini, D. D. Polarized monolayers formed by epithelial cells on a permeable and translucent support. J. Cell Biol. 77, 853–880 (1978).
-
Arthur, J. M. The MDCK cell line is made up of populations of cells with diverse resistive and transport properties. Tissue Cell 32, 446–450 (2000).
-
Furuse, M., Furuse, K., Sasaki, H. & Tsukita, S. Conversion of Zonulae Occludentes from Tight to Leaky Strand Type by Introducing Claudin-2 into Madin-Darby Canine Kidney I Cells. J. Cell Biol. 153, 263–272 (2001).
-
Green, I. J. Serial propagation of influenza B (Lee) virus in a transmissible line of canine kidney cells. Science 138, 42–43 (1962).
-
Brands, R., Visser, J., Medema, J., Palache, A. M. & van Scharrenburg, G. J. Influvac: a safe Madin Darby Canine Kidney (MDCK) cell culture-based influenza vaccine. Dev. Biol. Stand. 98, 93–100; discussion 111 (1999).
-
Matrosovich, M., Matrosovich, T., Carr, J., Roberts, N. A. & Klenk, H.-D. Overexpression of the α-2,6-Sialyltransferase in MDCK Cells Increases Influenza Virus Sensitivity to Neuraminidase Inhibitors. J. Virol. 77, 8418–8425 (2003).
