Cell Line Profile U-2 OS
(ECACC catalogue no. 92022711)
Cell Line History
U-2 OS is (Fig 1) one of the earliest described cancer derived cell lines. It was obtained from a moderately differentiated bone sarcoma of the tibia of a 15-year-old girl by Ponten and Saksela in 1964. The cell line was originally designated ‘2T’ and is believed to be one of the first human cell lines derived from a mesenchymal tumour1.
Key characteristics
The cell line U-2 OS is chromosomally aberrant with a polyploid karyotype. According to the literature the cell line secretes a platelet like derived growth factor (PDGF)-like protein2, expresses insulin-like growth factor (IGF) receptors and respond to the addition of IGF-I and II with an increase in DNA synthesis3.
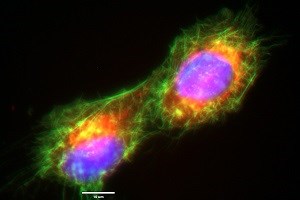
Figure 1: ECACC U-2 OS cells immediately following cell division showing actin cytoskeleton (phalloidin/Alexafluor488™ (green)), mitochondria (Mitotracker™ ROS (red)), and nuclei (NucBlue™ (blue) (all stains from Life Technologies)). Image captured by Gywain Jones using the EVOS-FL microscope with x100 objective under oil immersion.
STR Profile
|
Locus |
Score |
|---|---|
|
Amelogenin |
X |
|
CSF1P |
13 |
|
D13S317 |
13 |
|
D16S539 |
11,12 |
|
D5S818 |
11 |
|
D7S820 |
11,12 |
|
THO1 |
6,9.3 |
|
TPOX |
11,12 |
|
vWA |
14,18 |
Applications
The use of U-2 OS is widespread in the literature with thousands of Pubmed citations in many different fields and historically it has had a key application in bone formation, bone tumour and arthritis research4.
In more recent years, the cell line has shown itself to be useful as a successful transfection host using many commercially available kits, this combined with absence of any detectable adenovirus infection makes the cell line a useful expression platform in virology. For example: providing a suitable and more sensitive alternative to Huh-7 cells for siRNA studies on HCV RNA replication5 and in the study of the HPV11 genome6.
Although mesenchymal in origin, U-2 OS cells also have a flat, epithelial-like morphology making them a valuable tool in novel imaging technologies7. Examples of the U-2 OS cells’ use in imaging include: the development of fluorescent imaging techniques to examine dynamic events in live cells8 , in the technique of ‘Cell Painting’9 10, to assess cell proliferation in live cell imaging11 and in the detailed study of cell organelles such as mitochondria7 (Figure 2).
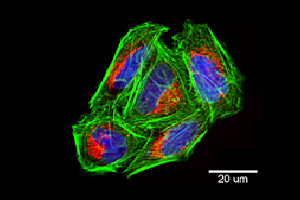
Figure 2: Confocal image of ECACC U-2 OS cells grown in multi-well plates showing individual mitochondria (Mitotracker™ ROS (red)), actin cytoskeleton (phalloidin/Alexafluor488™ (green)), and nuclei (NucBlue™ (blue) (all stains from Life Technologies). Image captured using the ImageXpress Micro Confocal High-Content Imaging System (Molecular Devices).
Culture Tips
Cells should be grown in tissue culture treated flasks in McCoy’s 5a medium supplemented with 1.5mM Glutamine and 10% foetal bovine serum (FBS). Cultures should be kept in a humidified 37°C / 5% CO2 incubator and the medium renewed two to three times a week.
Cells should be sub-cultured while still sub-confluent using Trypsin/EDTA solution following two washes with PBS (Ca2+ and Mg2+ free). Allow the flask to sit at room temperature (or at 37°C) until all the cells detach. Add fresh medium, aspirate and dispense into new culture flasks.
Related Cell Lines
|
ECACC Catalogue Number |
Cell Line Name |
Description |
|---|---|---|
|
Saos-2
|
Human primary osteogenic sarcoma from an 11 year-old female |
|
|
HOS |
Human osteosarcoma derived from a 13 year-old female. Cells are sensitive to both virus and chemical transformation and exhibit a flat morphology, low saturation density and low plating efficiency in soft agar. From the same patient as 143B (ECACC Catalogue number 91112502). |
|
|
143B |
Human bone osteosarcoma. Cells are thymidine kinase deficient (TK-) and resistant to BUdR. From the same patient as HOS (ECACC Catalogue number 87070202). |
|
|
KHOS-240S |
A revertant line of the KHOS/NP cell line (ECACC catalogue no. 84102903). Established after overnight incubation at 40.5°C and then sub-cloned at 36°C. The line has similar properties to the parent Human Osteogenic Cell Line HOS. Cells are non-tumourigenic in nude mice and no longer possess a rescuable MSV genome |
|
|
KHOS-312H |
A revertant line of the Human Osteogenic KHOS/NP cell line. Established after overnight incubation at 40.5°C and then sub-cloned at 36°C. The line has similar properties to the parent HOS cell line. Cells are non-tumourigenic in nude mice and no longer possess a rescuable MSV genome |
|
|
KHOS/NP |
Isolated from transformed foci of human osteogenic sarcoma (HOS) cells which were induced by Kirsten murine sarcoma virus. They contain MSV genome but do not produce infectious virus. The transformed cells exhibit a high saturation density, a high plating efficiency in soft agar and produce tumours in nude mice. |
|
|
MG-63 |
Human osteosarcoma from an osteosarcoma of a 14 year-old male. Produces high yields of interferon following superinduction with polyinosinic acid-polycytidilic acid, cyclohexamide and actinomycin. Antigenically, MG-63 interferon is more closely related to human fibroblast than to leukocyte interferon. |
References
-
Ponten, J. & Saksela, E. Two established in vitro cell lines from human mesenchymal tumours. Int. J. Cancer 2, 434–447 (1967).
-
Heldin, C. H. et al. A human osteosarcoma cell line secretes a growth factor structurally related to a homodimer of PDGF A-chains. Nature 319, 511–514 (1986).
-
Raile, K. et al. Human osteosarcoma (U-2 OS) cells express both insulin-like growth factor-I (IGF-I) receptors and insulin-like growth factor-II/mannose-6-phosphate (IGF-II/M6P) receptors and synthesize IGF-II: autocrine growth stimulation by IGF-II via the IGF-I receptor. J. Cell. Physiol. 159, 531–541 (1994).
-
Lauvrak, S. U. et al. Functional characterisation of osteosarcoma cell lines and identification of mRNAs and miRNAs associated with aggressive cancer phenotypes. Br. J. Cancer 109, 2228–2236 (2013).
-
Jones, D. M., Domingues, P., Targett-Adams, P. & McLauchlan, J. Comparison of U2OS and Huh-7 cells for identifying host factors that affect hepatitis C virus RNA replication. J. Gen. Virol. 91, 2238–2248 (2010).
-
Isok-Paas, H., Männik, A., Ustav, E. & Ustav, M. The transcription map of HPV11 in U2OS cells adequately reflects the initial and stable replication phases of the viral genome. Virol. J. 12, (2015).
-
Westrate, L. M., Drocco, J. A., Martin, K. R., Hlavacek, W. S. & MacKeigan, J. P. Mitochondrial Morphological Features Are Associated with Fission and Fusion Events. PLOS ONE 9, e95265 (2014).
-
Underwood, R. FRAP | Imaging & Microscopy - Research, Development, Production. Available at: https://www.imaging-git.com/applications/frap. (Accessed: 18th July 2019)
-
Bray, M.-A. et al. Cell Painting, a high-content image-based assay for morphological profiling using multiplexed fluorescent dyes. Nat. Protoc. 11, 1757–1774 (2016).
-
Bray, M.-A. et al. A dataset of images and morphological profiles of 30 000 small-molecule treatments using the Cell Painting assay. GigaScience 6, 1–5 (2017).
-
Musa, M., Kannan, T. P. & Mustafa, S. Cell Proliferation Study of Human Osteosarcoma Cell Line (U2OS) using Alamar Blue Assayand Live Cell Imaging. (2013). Available at: /paper/Cell-Proliferation-Study-of-Human-Osteosarcoma-Cell-Musa-Kannan/de32da3a8486b8f1d573dcaf09fb9b892139c0fc. (Accessed: 18th July 2019)
